 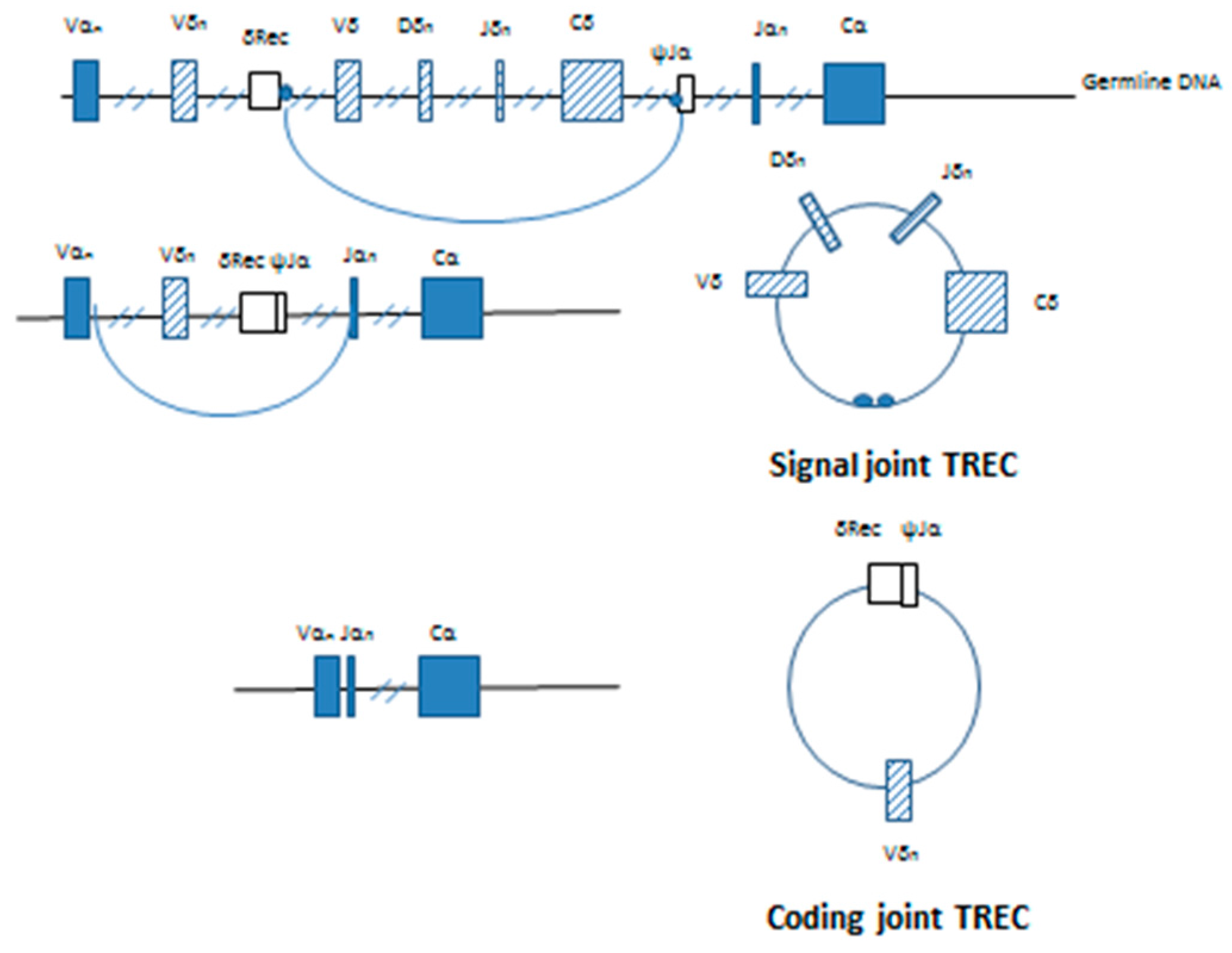
(iii) Information that demonstrates the performance characteristics of the test, including: The description must identify those control elements that are incorporated into the testing procedure. (F) A description of appropriate internal and external controls that are recommended or provided. Sample reports must include the scale used in reporting of results ( e.g., TREC copies/µL) and the range of values that will be reported out and (E) A description of the result outputs along with sample reports. The cutoff must be selected before conducting clinical and analytical studies (D) Methodology and protocols for detection of T-cell receptor excision circles and methods for determination of results. These specifications must include: descriptive characteristics of the filter paper, instructions on how a lab should choose the appropriate filter paper, chemical properties of the filter paper, interference concerns associated with the chemicals in the filter paper, absorption properties of the filter paper, punch size, absorption capacity, testing for homogeneity of punches, diameter of the circle for the dried blood spot aliquot, absorption time, physical composition, and number and size of punches to be tested (C) Specifications for the filter paper, which must be appropriately labeled for in vitro diagnostic use, to be used in specimen collection and how it will be used in specimen collection validation. (B) Detailed documentation of the device software including, but not limited to, standalone software applications and hardware-based devices that incorporate software (A) A detailed description of the test components, all required reagents, instrumentation and equipment, including illustrations or photographs of nonstandard equipment or methods (ii) A detailed description of all components in the test that includes: (B) The test is not intended to screen for less acute SCID syndromes, such as leaky SCID or variant SCID. (A) The test is not intended for diagnostic use, or for screening of SCID-like syndromes, such as DiGeorge syndrome or Omenn syndrome and (1) Premarket notification submissions must include the following information: The special controls for this device are: 
It is also not intended to screen for less acute SCID syndromes, such as leaky SCID or variant SCID. This test is not intended for use as a diagnostic test, or for screening of SCID-like syndromes, such as DiGeorge syndrome or Omenn syndrome. Presumptive positive results must be followed up by diagnostic confirmatory testing. A newborn screening test for SCID is a prescription device intended to measure T-cell receptor excision circle (TREC) DNA obtained from dried blood spot specimens on filter paper using a polymerase chain reaction based test as an aid in screening newborns for SCID. § 866.5930 Newborn screening test for severe combined immunodeficiency disorder (SCID). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
